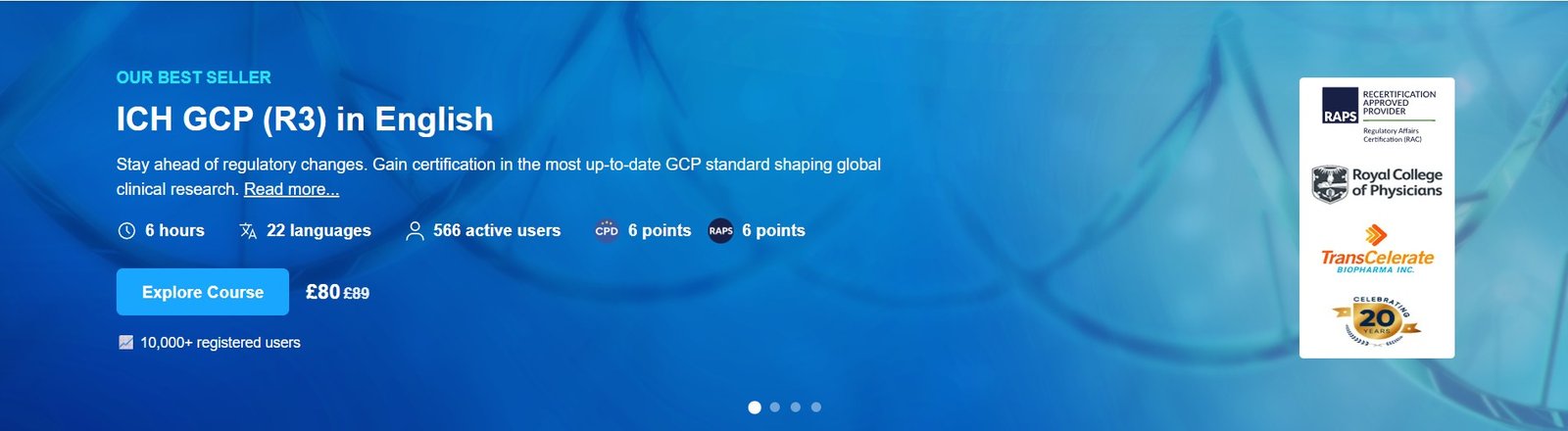
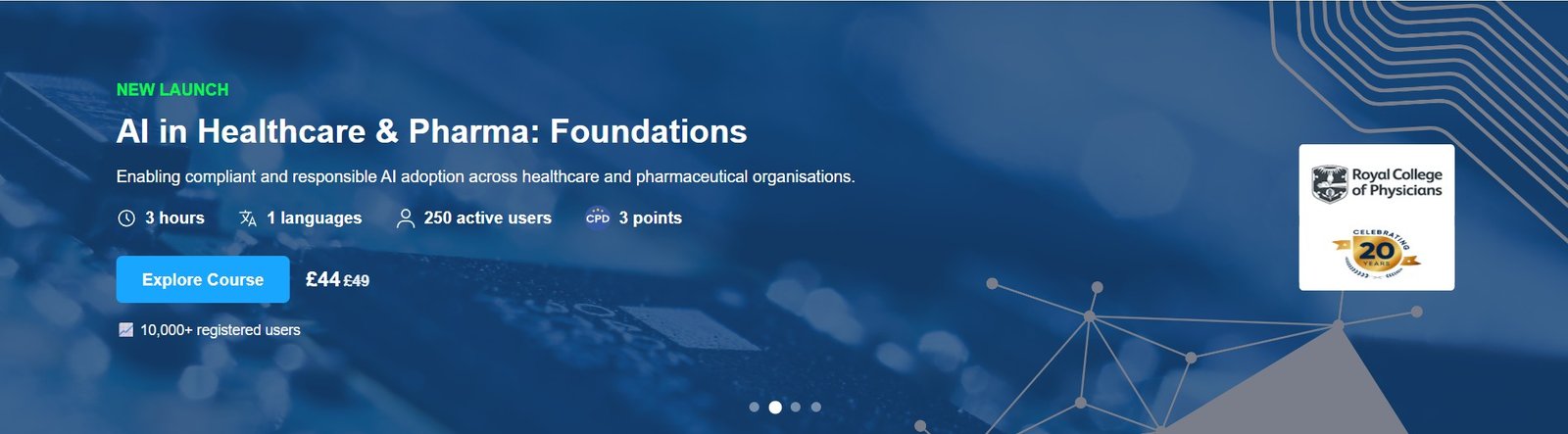
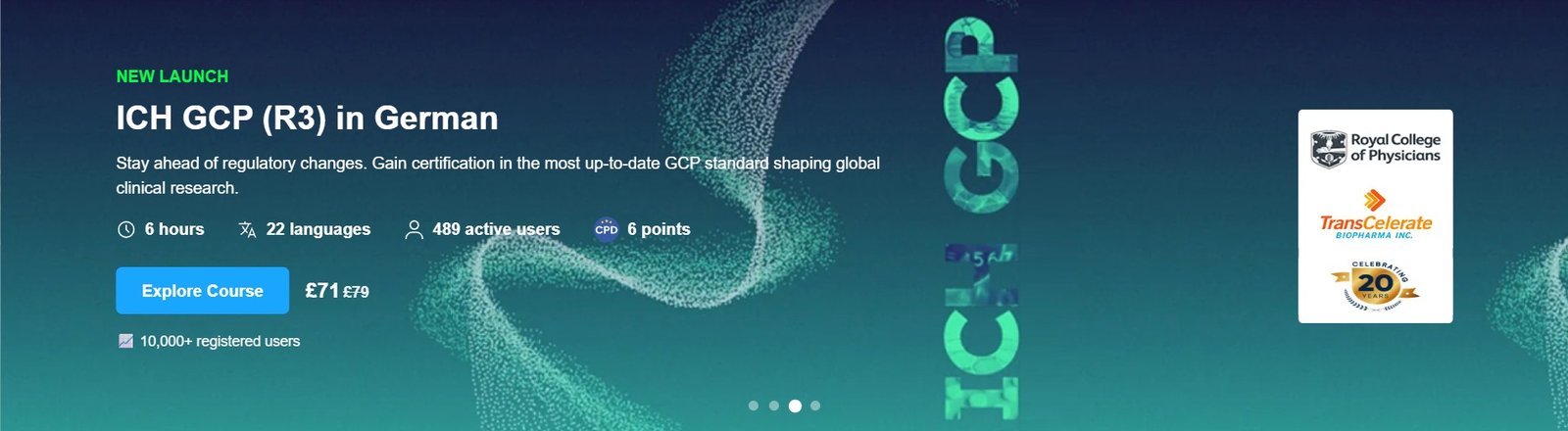
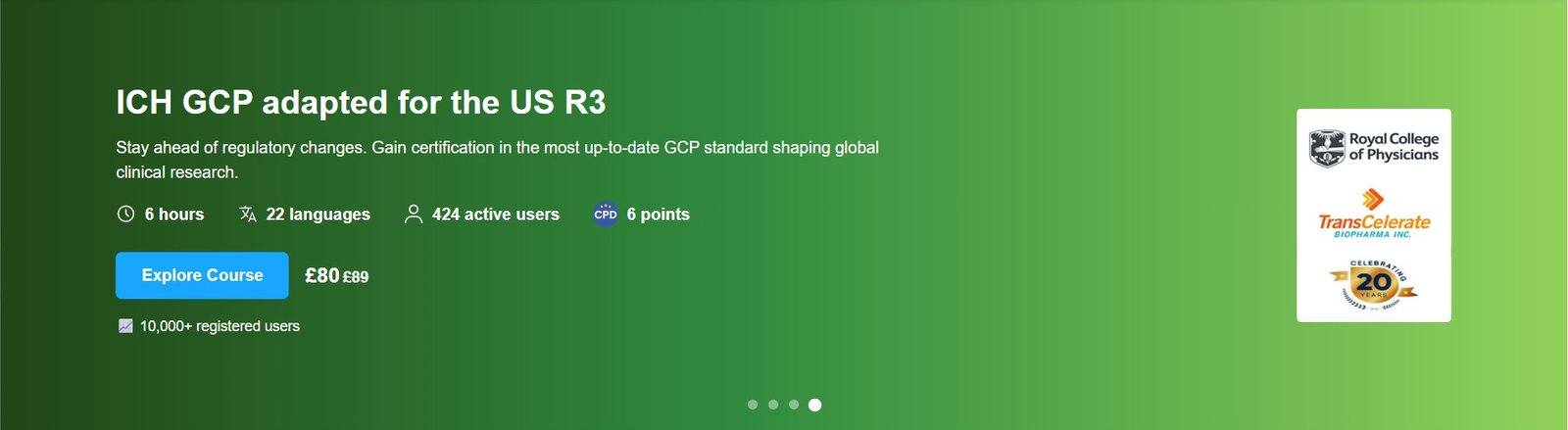
Course Overview
Hi, Welcome back!
- Led by experts with 25+ years in clinical research & regulatory oversight.
- Certification aligned with ICH-GCP, NDCT Rules 2019, and bioethics best practices.
- Trusted by investigators, ethics committees, and clinical professionals across India.
Testimonials

“The course explained GCP and bioethics concepts in a very simple and structured way. It helped me understand the fundamentals clearly.”

Anjali Verma
Clinical Research Student

“This training provided practical insights into clinical trial regulations and safety reporting. The modules were clear, informative, and very useful.”

Rahul Mehta
Clinical Research Professional
Limitless Learning, Limitless Possibilitie
- Lifetime Access
- Online Education
- Creative Learning
- Excellence Result
Call Us Now !
+91-9818884738

Frequently Asked Questions
What is GCP training and why is it important?
GCP (Good Clinical Practice) training provides essential knowledge about ethical and scientific standards used in clinical research. It ensures that clinical trials are conducted safely, participants’ rights are protected, and research data is reliable and accurate.
Who should take this course?
This course is designed for clinical researchers, doctors, investigators, study coordinators, healthcare professionals, medical students, and anyone involved in clinical trials or research activities.
Is the course certified or accredited?
Yes, the course provides a certificate upon successful completion. The certificate confirms that the learner has completed GCP training and understands the basic principles of Good Clinical Practice.
How long does it take to complete the course?
The course duration typically ranges from a few hours to a few days depending on the modules included. Most learners can complete the course at their own pace.
Are there assessments or evaluations?
Yes, the course includes assessments or quizzes at the end of modules to evaluate understanding. Learners must successfully complete these assessments to receive the course certificate.
Address
166-B, First Floor, Pocket AG-1, Vikaspuri, Delhi-110018
shodhclinical01@gmail.com
shodhclinical05@gmail.com
Our Solutions
Corporate Training
Meet the Experts
Dr Monika Bahl
Copyright © 2026 shodhclinicals.org | All right reserved